Welcome to Standphill India
BIS Certification for Medical Textiles in India
ISI Mark Licence under the Medical Textiles (Quality Control) Order, 2023
Quick Answer — BIS Certification for Medical Textiles
BIS certification is mandatory for medical textiles in India under the Medical Textiles (Quality Control) Order, 2023 issued by the Ministry of Textiles. All specified medical textile products must carry the ISI Mark to ensure safety and quality. Manufacturers must comply with standards for products like Surgical Masks (IS 16289:2014), Surgical Gowns (IS 17334:2019), Baby Diapers (IS 17509:2021), Sanitary Napkins (IS 5405:2019), and 50 other medical textile standards. The process involves application, factory audit, and sample testing in BIS-approved laboratories. The compliance deadline under the Medical Textiles (Quality Control) Second Amendment Order, 2025 is 31 December 2025. Standphill India provides complete BIS medical textile certification consultancy across India with over 20 years of experience.
Overview
BIS Certification for Medical Textiles is a mandatory product certification issued by the Bureau of Indian Standards (BIS) for all specified medical textile products sold, manufactured, or imported in India. Medical textiles play an essential protective function in the healthcare industry — from high-volume disposable products like baby diapers, sanitary napkins, and surgical face masks to highly specialized products like dressings, bandages, surgical gowns, and bio-protective coveralls.
Because medical textile products come into direct contact with the human body — often in clinical, surgical, or hygiene-sensitive settings — ensuring their quality, safety, and performance is critical for protecting the health of patients, healthcare workers, and consumers. The Medical Textiles (Quality Control) Order, 2023 issued by the Ministry of Textiles makes BIS certification with the ISI Mark mandatory for all specified medical textile products before they can be manufactured, imported, or sold in India.
The compliance deadline has been extended to 31 December 2025 under the Medical Textiles (Quality Control) Second Amendment Order, 2025. All medical textile manufacturers and importers must obtain BIS certification before this deadline to avoid product seizure, import rejection, and legal penalties.
Standphill India provides end-to-end BIS certification consultancy for medical textile manufacturers across India, covering documentation preparation, product testing coordination, factory inspection preparation, and ISI Mark licence approval support.
⚠️ Important Compliance Deadline
The Medical Textiles (Quality Control) Second Amendment Order, 2025 has extended the compliance deadline to 31 December 2025. All medical textile manufacturers and importers must obtain BIS certification before this date. Products without the ISI Mark after this deadline cannot be legally manufactured, imported, or sold in India. Contact Standphill India immediately to begin your certification process.
What is BIS Certification for Medical Textiles?
BIS Certification for Medical Textiles is the mandatory product certification system under which medical textile manufacturers must demonstrate that their products comply with the applicable Indian Standards before placing them in the Indian market.
The Bureau of Indian Standards (BIS) is India's national standards body responsible for standard formulation, marking, and quality certification of goods. Under the BIS product certification scheme — also known as the ISI Mark Certification Scheme — BIS grants a licence to manufacturers to use the ISI Mark on certified products.
When a medical textile product carries the ISI Mark, it provides third-party assurance to healthcare providers, patients, hospitals, and consumers that the product meets all the requirements of the applicable Indian Standard — including performance, safety, hygiene, and quality benchmarks.
Without a valid ISI Mark licence, medical textile products covered under the QCO 2023 cannot be legally manufactured, imported, sold, supplied, or distributed in India after the compliance deadline of 31 December 2025.
Medical Textiles Quality Control Order (QCO), 2023
The Medical Textiles (Quality Control) Order, 2023 was issued by the Ministry of Textiles, Government of India, to regulate the quality and safety of medical textile products sold in India. It mandates compliance with specified Indian Standards and makes the ISI Mark mandatory for all covered medical textile products.
The QCO covers a wide range of medical textile products including dressings, bandages, adhesive plasters, surgical masks, surgical gowns, baby diapers, sanitary napkins, shoe covers, caps, bed sheets, bio-protective coveralls, compression stockings, and many other products.
The QCO was issued to achieve the following objectives:
• ensure that medical textile products sold in India meet minimum safety and quality standards
• protect the health of patients, healthcare workers, and consumers from substandard medical textiles
• align Indian medical textile standards with global quality norms
• prevent the circulation of unsafe and non-compliant medical textile products in the Indian market
• build consumer and institutional trust in Indian medical textile products
The Medical Textiles (Quality Control) Second Amendment Order, 2025 has given manufacturers and importers a final grace period until 31 December 2025 to achieve full compliance. After this date, non-certified products face seizure and legal action.
Why is BIS Certification Important for Medical Textiles?
1. Legal Compliance and Market Access
Medical textile products covered under the QCO 2023 cannot be legally manufactured, imported, or sold in India without a valid ISI Mark licence after 31 December 2025. BIS certification is the mandatory gateway to legal market access.
2. Patient and Consumer Safety
Medical textiles are used in direct contact with the human body — often in surgical, clinical, or hygiene-sensitive settings. BIS certification ensures that these products meet stringent safety and performance standards, reducing the risk of infections, allergic reactions, or product failure that could harm patients and users.
3. Product Quality Assurance
The ISI Mark on medical textiles confirms that the product has been tested and evaluated against the applicable Indian Standard. This gives healthcare providers, hospitals, institutional buyers, and consumers confidence in the product's quality and reliability.
4. Prevention of Substandard Products
The certification process involves detailed testing by BIS recognized laboratories to verify that medical textile products meet performance requirements. This effectively weeds out substandard or unsafe products from the Indian market.
5. Import Clearance
Customs authorities in India verify BIS certification for regulated medical textile products at ports of entry. Products without the ISI Mark are rejected or detained at customs. Certification ensures smooth import clearance.
6. International Recognition
BIS certification for medical textiles helps Indian manufacturers meet quality standards that are aligned with international benchmarks, supporting export credibility and access to global markets.
Who Needs BIS Certification for Medical Textiles?
BIS certification under the Medical Textiles QCO 2023 is required for:
• Indian manufacturers producing specified medical textile products at factories in India
• Foreign manufacturers exporting specified medical textile products to India (under the FMCS scheme)
• Importers and distributors dealing in medical textile products covered under the QCO 2023
The ISI Mark licence is issued only to the manufacturer of the product. Foreign manufacturers must appoint an Authorized Indian Representative (AIR) to manage regulatory communication and compliance formalities with BIS in India.
Separate BIS licences are required for each product type and each manufacturing location.
Also Read
Key Mandatory Indian Standards for Medical Textiles – BIS Certification
These are the most commonly required BIS certifications for medical textile products under the QCO 2023. Each product must comply with its specific Indian Standard before obtaining the ISI Mark licence.
| Product | Indian Standard |
|---|---|
| Surgical Face Masks | IS 16289:2014 |
| Surgical Gowns and Surgical Drapes | IS 17334:2019 |
| Bio-Protective Coveralls | IS 17423:2021 |
| Sanitary Napkins | IS 5405:2019 |
| Disposable Baby Diaper | IS 17509:2021 |
| Reusable Sanitary Pad / Period Panties | IS 17514:2021 |
| Shoe Covers | IS 17349:2020 |
| Caps | IS 17629:2021 |
| Bed Sheet and Pillow Cover | IS 17630:2021 |
| Disposable Adult Incontinence Diaper | IS 17508:2020 |
| Eye Pad | IS 17628:2021 |
| Abdominal Binder | IS 17350:2020 |
| Pressure Garment | IS 17353:2020 |
| Dental Bib / Napkins | IS 17354:2020 |
| Adhesive Incise Drape | IS 17348:2020 |
| Foam Dressing | IS 17352:2020 |
| Hydrocolloid Dressing | IS 17506:2020 |
| Anti-Embolic Stocking | IS 17359:2020 |
| Cellulose Wading | IS 17507:2020 |
| Chlorhexidine Gauze Dressings | IS 17528:2021 |
In addition to the above, BIS certification covers the following medical textile categories under the QCO 2023:
• Elastic Adhesive Dressing (IS 16669:2017)
• Medicated Paraffin Gauze Dressings (IS 16291:2014)
• Knitted Viscose Primary Dressings (IS 16290:2014)
• Providone Iodine Ointment Based Knitted Dressing (IS 16466:2016)
• Absorbent Cotton – Sterile and Non-Sterile (IS 16468:2016)
• Elastic Surgical Adhesive Tapes (IS 16470:2016)
• Absorbent Cotton Ribbon Gauze (IS 16670:2017)
• Salicylic Acid Adhesive Plaster (IS 16668:2017)
• Permeable Nonwoven Surgical Adhesive Tape (IS 16948:2018)
• Xray Detectable Absorbent Cotton Gauze (IS 16950:2018)
• Handloom Cotton Lint, Absorbent, Bleached (IS 757:1971)
• Handloom Cotton Gauze, Absorbent, Non-Sterilized (IS 758:1988)
• Handloom Cotton Bandage Cloth, Non-Sterilized (IS 863:1988)
• Xray Detectable Gauze Swabs and Laparotomy Sponges (IS 10829:1993)
• Crepe Bandage (IS 4605:1981)
• Open Weave Bandages (IS 16469:2016)
• Elasticated Tubular Bandages (IS 16946:2018)
• Elastic Bandage (IS 16111:2013)
• Non-Woven Bandage Rolls (IS 16660:2017)
• Orthopedic Stockinet (IS 16302:2014)
• Cast Padding for Orthopedic Plaster (IS 16303:2014)
• Graduated Medical Compression Stockings (IS 16467:2016)
• Zinc Oxide Elastic Self-Adhesive Bandage (IS 4739:1986)
• Zinc Oxide Self-Adhesive Plaster (IS 4717:1980)
• Bandage, Plaster of Paris (IS 4738:1993)
• First Aid Dressings (IS 11163:1985)
• Dressing, Shell Compressed (IS 17351:2020)
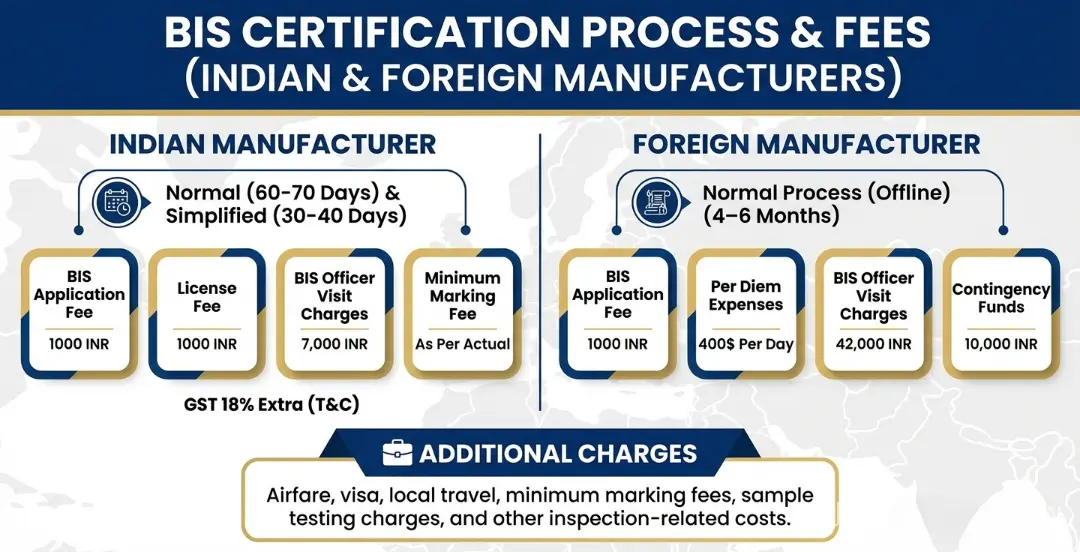
BIS Certification Process for Medical Textiles
Step 1 – Documentation Preparation
The manufacturer prepares all required technical and business documents before application submission. This includes factory registration details, manufacturing process flow chart, quality control plan, in-house testing facility details, raw material specifications, and product technical specifications as per the applicable Indian Standard.
Step 2 – Application Submission
The manufacturer submits the application through the BIS ManakOnline portal along with all supporting documents. Foreign manufacturers submit a physical application to the BIS Foreign Manufacturers division and must appoint an Authorized Indian Representative (AIR). The application fee and 50% of the minimum marking fee are paid at this stage.
Step 3 – Scrutiny of Application by BIS
BIS officials carefully review each document and the application submitted. If any information is missing or clarification is needed, BIS raises queries through the portal. The manufacturer must respond promptly to avoid delays.
Step 4 – Factory Audit by BIS Auditor
BIS appoints an auditor to inspect the manufacturing premises. During the factory audit, the BIS auditor inspects:
• manufacturing infrastructure and production machinery
• in-house testing facilities and calibrated testing equipment
• quality control systems, production records, and process documentation
• raw material controls and storage conditions
During the audit, the BIS auditor draws product samples and sends them to a BIS-approved laboratory for independent testing.
Step 5 – Sample Testing in BIS Approved Laboratory
All product samples collected during the factory audit are tested in a BIS-approved laboratory to verify that the medical textile product meets all the requirements of the applicable Indian Standard — including performance, safety, hygiene, and physical properties.
Step 6 – Grant of BIS Licence
If the product complies with all the specified Indian Standard requirements and the factory audit is satisfactory, BIS grants the ISI Mark licence. The manufacturer is then authorized to use the ISI Mark on certified medical textile products. The BIS licence is valid for two years and must be renewed to maintain certification validity.
BIS Certification Process for Medical Textiles
Documents Required for BIS Certification of Medical Textiles
Manufacturers must prepare and submit the following documents during the BIS certification application process. Incomplete documentation is the most common cause of BIS certification delays.
• factory registration certificate or manufacturing licence
• company registration documents (GST certificate, company incorporation certificate)
• manufacturing process flow chart
• quality control plan and parameters
• factory layout plan and equipment details
• list of manufacturing machinery with specifications
• list of in-house testing equipment with calibration certificates
• laboratory test reports from BIS-approved laboratories
• product specifications and technical details as per applicable Indian Standard
• raw material specifications and supplier details
• declaration of conformity to the applicable Indian Standard
• authorized signatory details and board resolution or authorization letter
• identity proof of authorized signatory
• AIR documents (mandatory for foreign manufacturers)
Standphill India assists in documentation preparation, scrutiny, and filing support to improve approval efficiency and reduce delays at every stage of the certification process.
Also Read
BIS Certification for Indian Manufacturers (ISI Mark – Scheme I)
BIS Certification Fees for Medical Textiles
The cost of BIS medical textile certification includes the following standard fee components:
| S. No. | Particulars | Amount |
|---|---|---|
| 1 | APPLICATION FEE | INR 1,000/- |
| 2 | PRELIMINARY INSPECTION CHARGES | INR 7,000/- |
| 3 | ANNUAL LICENCE FEE | INR 1,000/- |
| 4 | MINIMUM MARKING FEE (Micro Scale 80% Off, Small 50%, Medium 20%) |
As Applicable |
| 5 | Sample Testing Fee (BIS Approved Lab) | As Applicable |
Additional costs may include investment in factory facilities, particularly for setting up or upgrading in-house testing laboratories as required under the BIS Scheme of Inspection and Testing for the applicable medical textile standard. The exact total cost varies by product type and standard.
Timeline for BIS Medical Textile Certification
The BIS certification process for medical textiles typically takes 30 to 60 days for Indian manufacturers.
The timeline depends on:
• completeness of documentation at application stage
• laboratory testing schedule at BIS-approved labs
• factory inspection scheduling by BIS
• response time to BIS queries if raised
• number of product types being certified
Given the compliance deadline of 31 December 2025, manufacturers should begin the certification process immediately to avoid last-minute delays and ensure timely approval before the deadline.
Surveillance and Licence Renewal
BIS medical textile certification does not end with the grant of licence. After approval, BIS conducts periodic surveillance to ensure the manufacturer continues to comply with licence conditions. The BIS medical textile licence is valid for two years and must be renewed periodically.
Surveillance activities may include:
• factory inspections
• market sample testing of ISI-marked medical textile products
• review of production records, quality control documentation, and in-house testing logs
Failure to maintain compliance or renew the licence may result in suspension or cancellation, which would require the manufacturer to cease use of the ISI Mark immediately.
Recent BIS Medical Textile Certifications Delivered by Standphill India
Standphill India has successfully assisted medical textile manufacturers in obtaining BIS ISI Mark certification.
Shree Nagai Enterprises
Products Certified
Medical Textiles – Surgical Face Masks
Specification as per IS 16289:2014
Medical Textiles – Shoe Covers
Specification as per IS 17349:2020
This project demonstrates Standphill India's expertise in handling BIS certification for medical textile products including product testing coordination, documentation preparation, and ISI Mark licence approval support.
Why Choose Standphill India for BIS Medical Textile Certification?
Standphill India is a leading BIS certification consultancy with over 20 years of experience and a proven track record of 10,000+ domestic and 400+ foreign product certifications completed across India and worldwide.
Our specific services for BIS medical textile certification include:
• identification of the applicable Indian Standard for your specific medical textile product
• complete documentation preparation including manufacturing flow chart, QC plan, and technical specifications
• coordination with BIS-approved laboratories for medical textile product testing
• factory inspection preparation and in-house testing facility guidance
• BIS application filing on ManakOnline portal
• BIS query resolution and regulatory communication management
• ISI Mark licence approval support and post-licence renewal assistance
• FMCS application support for foreign medical textile manufacturers
• urgent certification support for manufacturers approaching the 31 December 2025 deadline
Conclusion
BIS Certification for Medical Textiles is a mandatory legal and safety compliance requirement for all medical textile manufacturers, importers, and sellers in India under the Medical Textiles (Quality Control) Order, 2023. With the compliance deadline of 31 December 2025 under the Medical Textiles (Quality Control) Second Amendment Order, 2025, manufacturers must act immediately to obtain BIS certification before this date.
Medical textiles — from surgical face masks and baby diapers to bandages, dressings, and surgical gowns — are products that directly affect the health and safety of patients, healthcare workers, and consumers. BIS certification with the ISI Mark ensures these products meet rigorous Indian quality and safety standards before reaching the market.
From application preparation and laboratory testing to factory inspection and ISI Mark licence approval, the certification process requires proper planning, documentation, and technical understanding of the applicable Indian Standards.
With Standphill India as your certification partner, medical textile manufacturers can complete the BIS certification journey with stronger compliance support, smoother execution, and full confidence — before the deadline.
Get expert support for BIS certification for medical textiles and ensure your products are certified, compliant, and market-ready before 31 December 2025.
Frequently Asked Questions (FAQ)
Q1. Is BIS certification mandatory for medical textiles in India?
Yes. BIS certification is mandatory for all specified medical textile products in India under the Medical Textiles (Quality Control) Order, 2023. Products must carry the ISI Mark before they can be manufactured, imported, or sold in India. The compliance deadline is 31 December 2025 under the Medical Textiles (Quality Control) Second Amendment Order, 2025.
Q2. What is the Medical Textiles Quality Control Order 2023?
The Medical Textiles (Quality Control) Order, 2023 was issued by the Ministry of Textiles, Government of India, making BIS certification mandatory for specified medical textile products. It covers 54 Indian Standards for products including surgical masks, baby diapers, sanitary napkins, bandages, dressings, surgical gowns, shoe covers, caps, and many more medical textile products.
Q3. What is the compliance deadline for medical textile BIS certification?
The compliance deadline under the Medical Textiles (Quality Control) Second Amendment Order, 2025 is 31 December 2025. All manufacturers and importers of covered medical textile products must obtain BIS certification before this date. Products without the ISI Mark after this deadline will face seizure, import rejection, and legal action.
Q4. Which medical textile products require BIS certification?
BIS certification is required for a wide range of medical textile products including surgical face masks (IS 16289:2014), surgical gowns and drapes (IS 17334:2019), bio-protective coveralls (IS 17423:2021), sanitary napkins (IS 5405:2019), disposable baby diapers (IS 17509:2021), reusable sanitary pads and period panties (IS 17514:2021), shoe covers (IS 17349:2020), caps (IS 17629:2021), bandages, dressings, adhesive plasters, and 40 other medical textile standards.
Q5. What is the process for BIS certification of medical textiles?
The BIS certification process for medical textiles involves documentation preparation, application submission on BIS ManakOnline portal, BIS scrutiny of application, factory audit by BIS-appointed auditor, sample testing in BIS-approved laboratory, and grant of ISI Mark licence. The licence is valid for two years and must be renewed. Standphill India provides end-to-end support for all steps.
Q6. How long does BIS certification for medical textiles take?
BIS certification for medical textiles typically takes 30 to 60 days for Indian manufacturers depending on documentation readiness, laboratory testing schedules, and factory inspection availability. Given the 31 December 2025 deadline, manufacturers should begin the process immediately.
Q7. What is the cost of BIS certification for medical textiles?
Government fees include application fee INR 1,000, preliminary inspection charges INR 7,000, and annual licence fee INR 1,000. Additional costs include laboratory testing charges from BIS-approved labs and marking fees as applicable. Investment in in-house testing facilities may also be required. Contact Standphill India for a personalized cost estimate for your specific medical textile product.
Q8. Can foreign manufacturers get BIS certification for medical textiles?
Yes. Foreign manufacturers must apply under the BIS Foreign Manufacturers Certification Scheme (FMCS) and appoint an Authorized Indian Representative (AIR) in India. The Indian Standards, testing requirements, and certification process are the same as for Indian manufacturers.
Q9. What documents are required for BIS certification of medical textiles?
Key documents include factory registration certificate, manufacturing process flow chart, quality control plan, laboratory test reports from BIS-approved labs, factory layout and equipment details, company registration proof, product specifications, declaration of conformity to Indian standards, in-house testing facility details, and authorized signatory documents. Foreign manufacturers must also provide AIR appointment documents.
Q10. Does Standphill India provide BIS medical textile certification support across India?
Yes. Standphill India has a strong presence across all States and Union Territories of India and supports medical textile manufacturers nationwide with complete BIS certification consultancy under the Medical Textiles QCO 2023 — from standard identification and documentation to factory inspection preparation and ISI Mark licence approval. We have successfully certified medical textile products including Surgical Face Masks (IS 16289:2014) and Shoe Covers (IS 17349:2020) for manufacturers across India.
Get Expert BIS Certification Support for Your Business
Connect with Standphill India for complete support with BIS certification, CRS registration, WPC approval, FMCS compliance, and related regulatory services. Our team helps businesses with documentation, testing coordination, application support, and end-to-end compliance guidance.
Request Free Consultation
Share your requirement and our team will contact you shortly.





